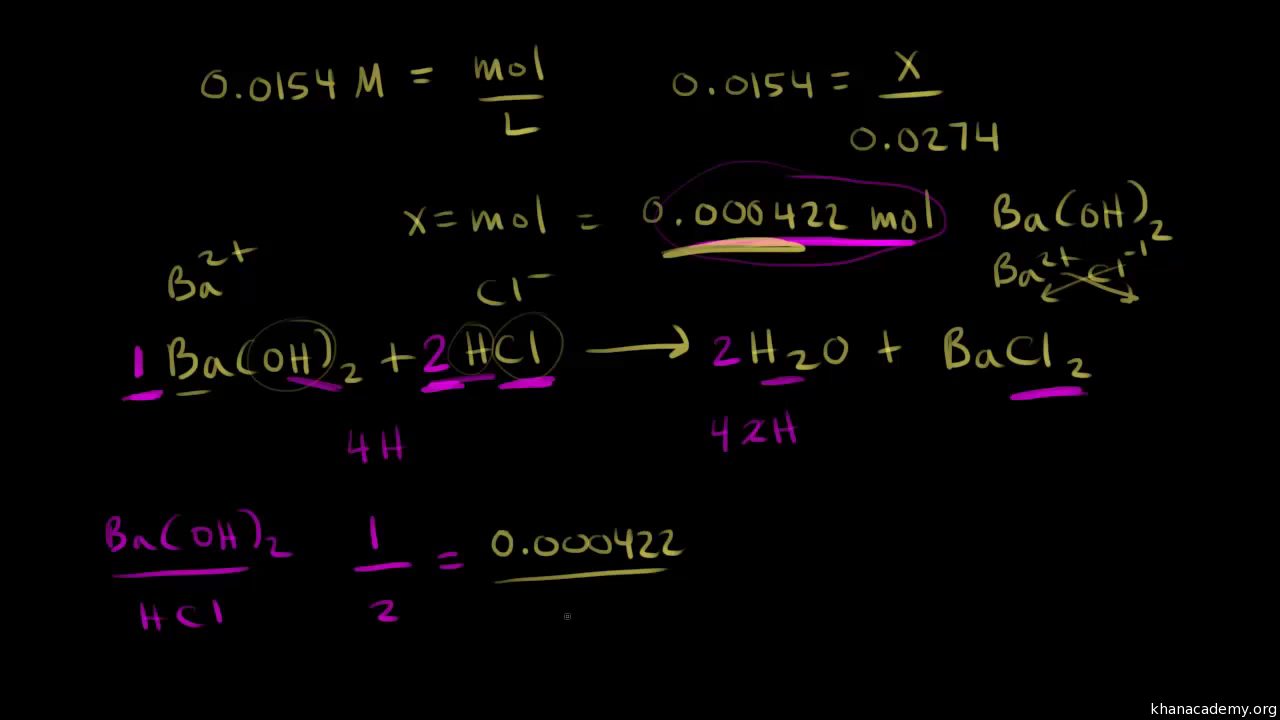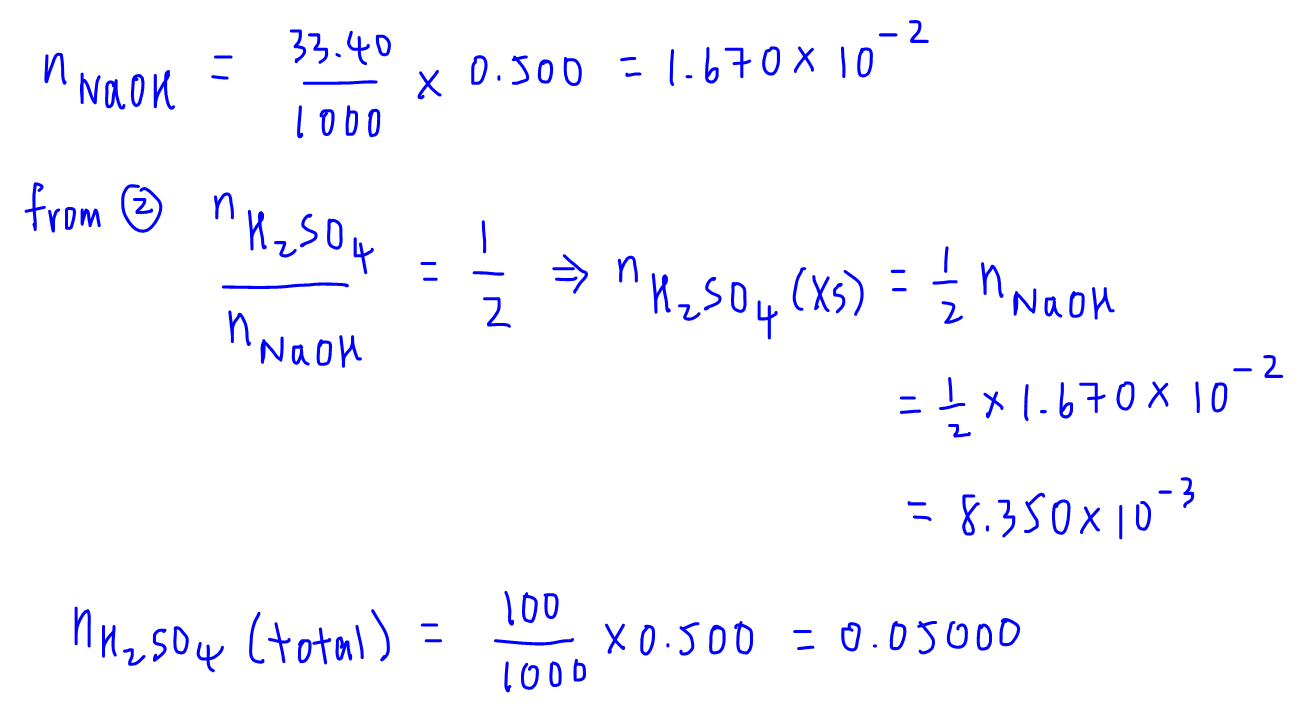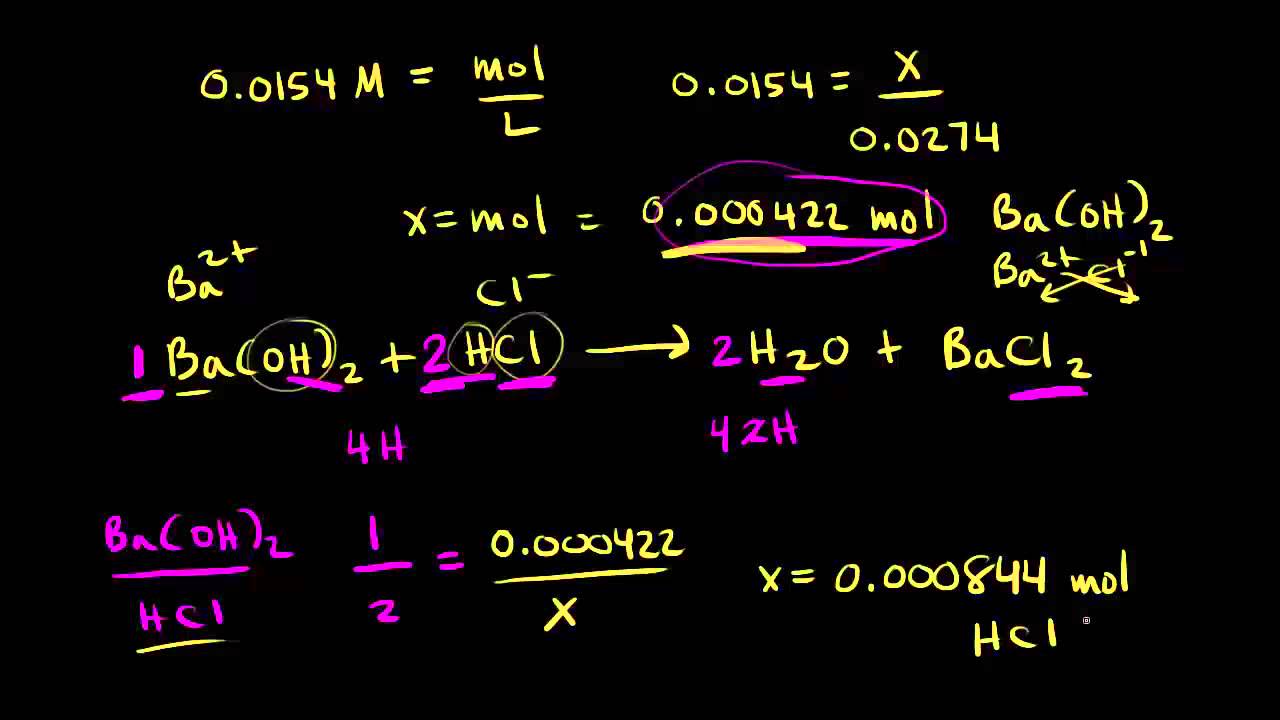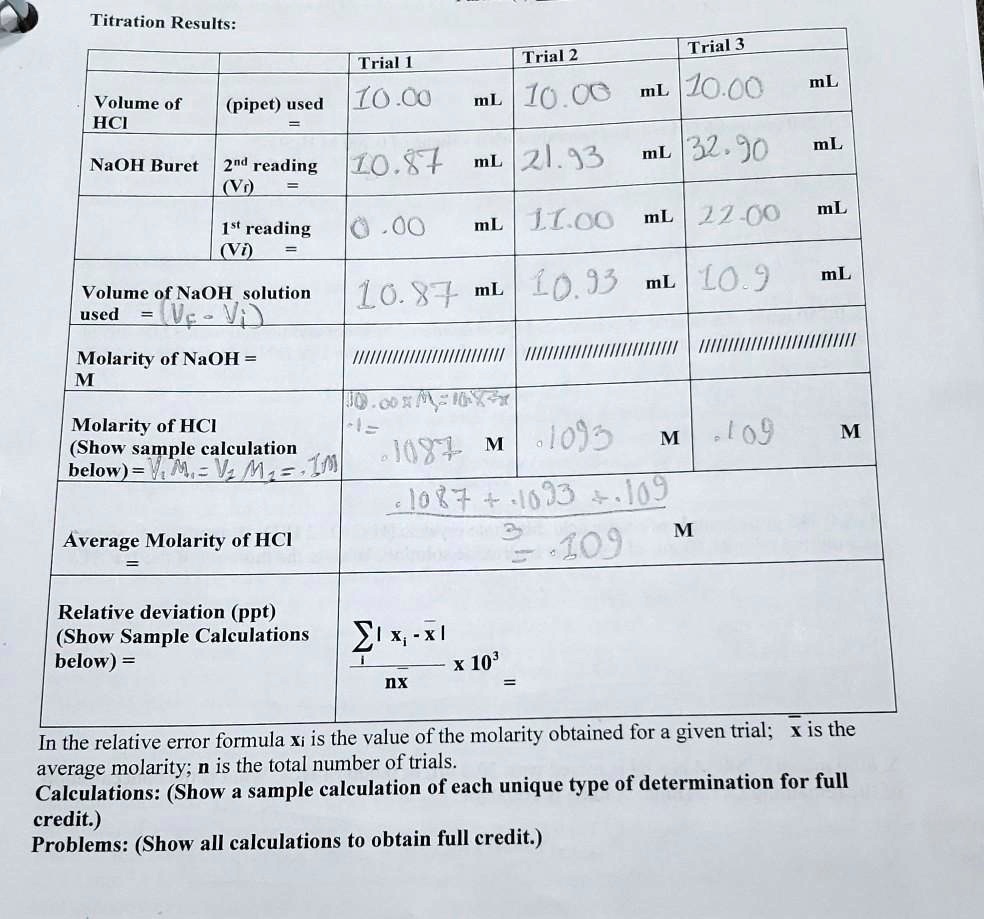
SOLVED: Titration Results: Trial 3 Trial 2 1o CO mL Jo.co mL Trial [ Volume of HCI (pipet) used Lo C NaOH Buret 2nd reading ILO;` 87 mL 21.93 mL 32 .

Acid Base Titration Problems, Basic Introduction, Calculations, Examples, Solution Stoichiometry - YouTube
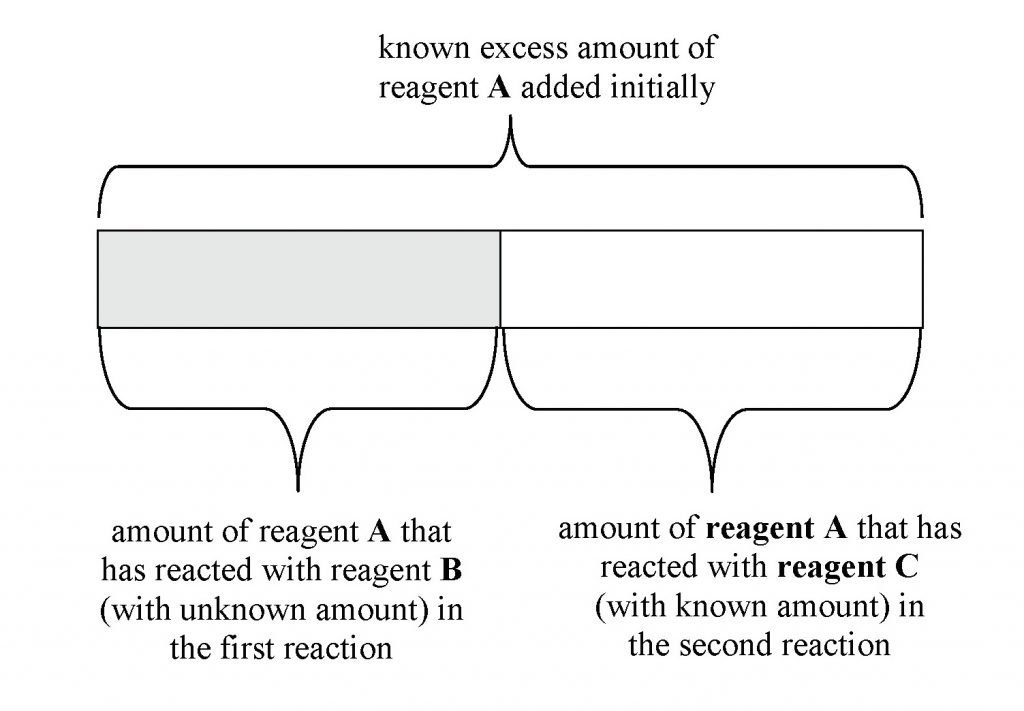
5 Simple Steps to Solve Back Titration Questions in Volumetric Analysis - A-Level H2 Chemistry Tuition by 10 Year Series Author

SOLVED: REPORT SHEET Acid-Base Titration LAB 10 Concentration - of Acetic Acid in Vinegar inee Volume Brand 5.0 mL (90 on label) OfNOH 2oz Molarity (M) Trial Trial 2 Trial 3 55

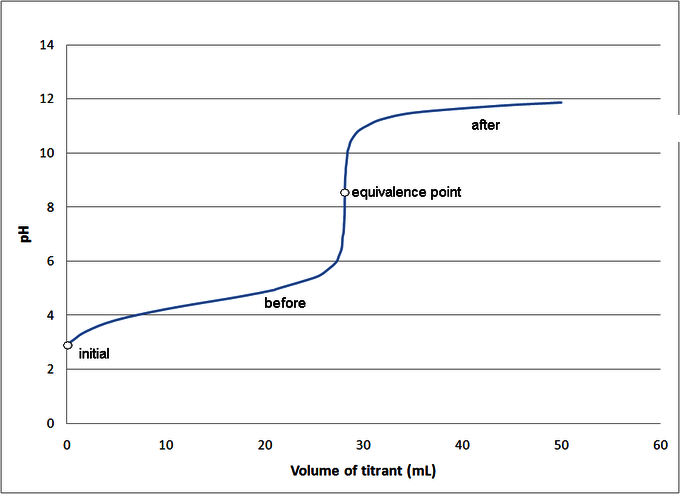
How to Calculate Analyte Concentration Using the Equivalence Point in an Acid-base Titration | Chemistry | Study.com