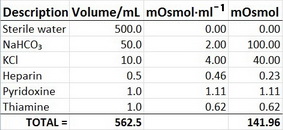
What is the molarity (M) of a 5% dextrose (IV sugar solution) solution? (hint: molarity= number of moles of solute in 1 liter of 1000mL of solution). Show your calculations. | Homework.Study.com

Calculate your own osmolarity | Lab values and concentrations | Health & Medicine | Khan Academy - YouTube

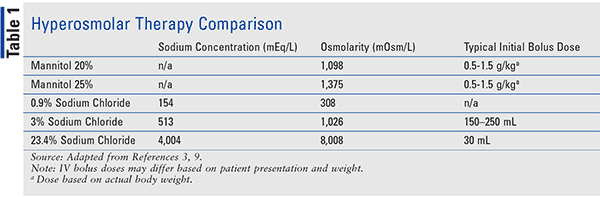
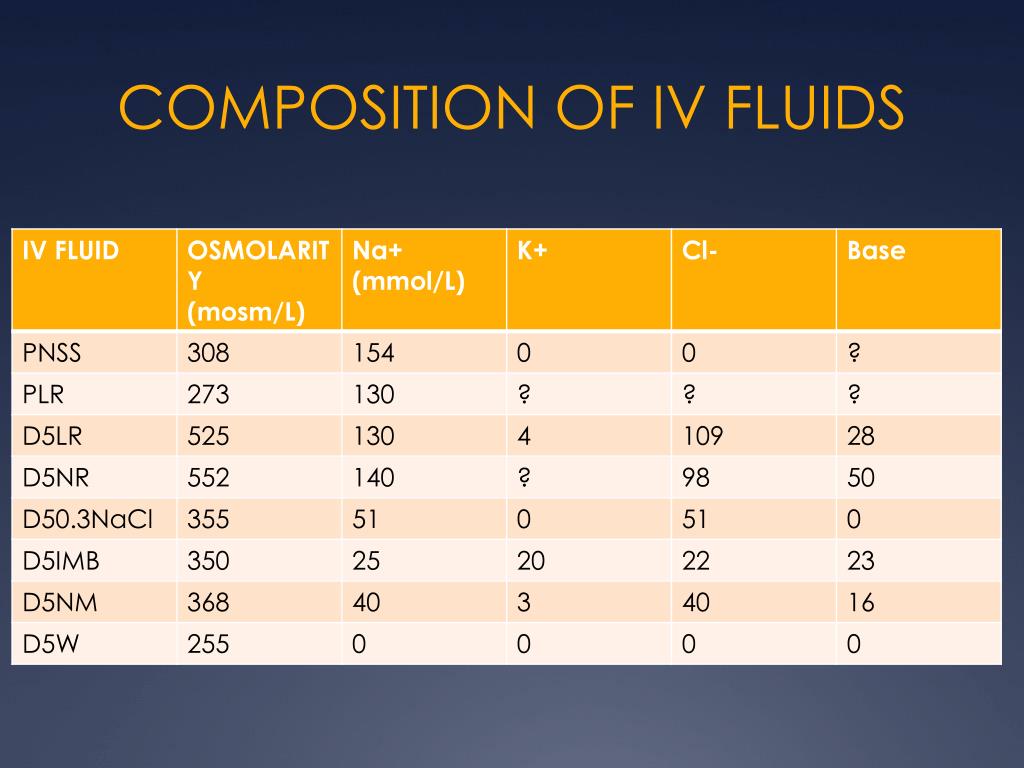
تويتر \ ph.jawzaa|ص.جوزاء على تويتر: "Example 1: Calculate the ideal osmolarity of 0.9 % NaCl injection? Because of bonding forces, however, n is slightly less than 2 for solutions of sodium chloride